Sf2 dipole moment7/12/2023 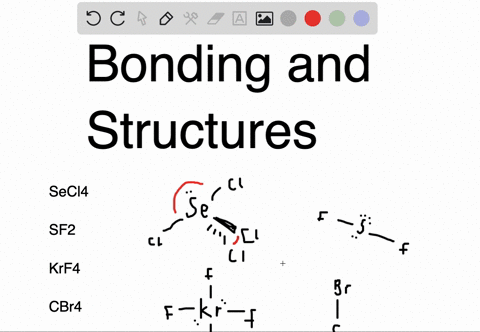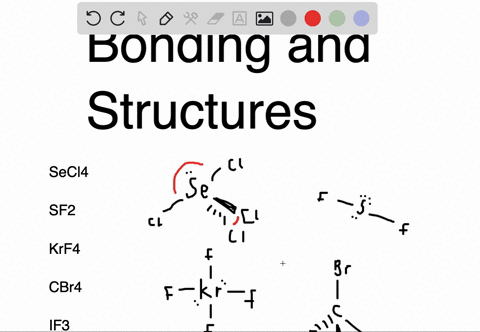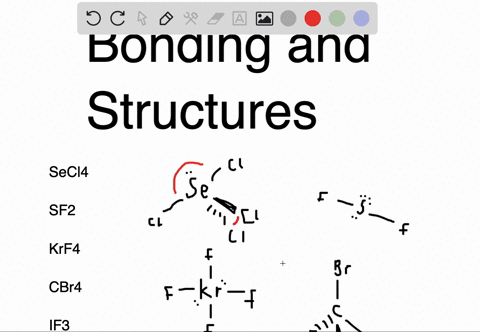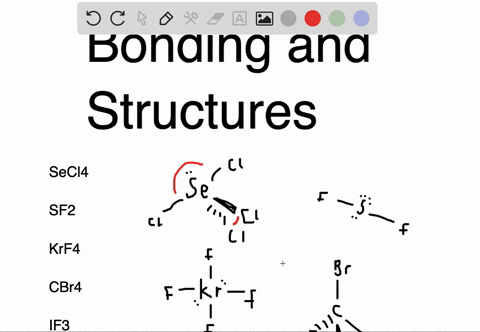 
Elements up to period four on the periodic table need eight electrons to fill their outer electron shell. 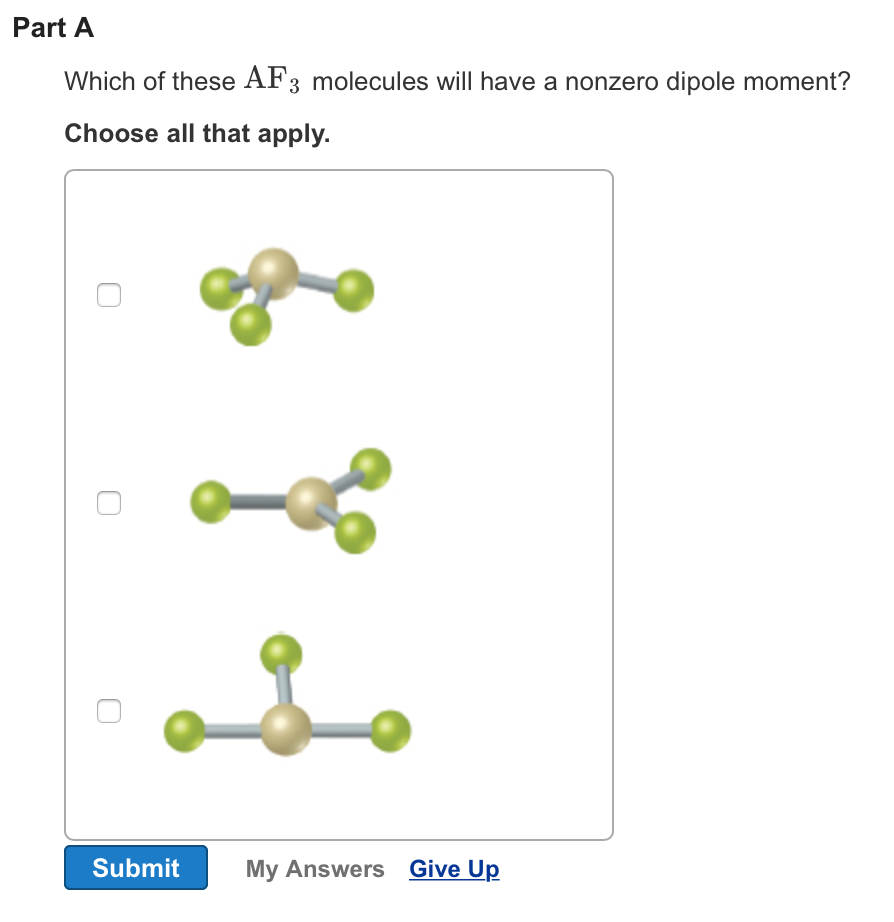
An atom is considered "happy" when its outer electron shell is filled. These instructions outline the Kelter strategy to draw Lewis structures for molecules. Chemistry students are often confused by the models, but drawing Lewis structures can be a straightforward process if the proper steps are followed.īe aware there are several different strategies for constructing Lewis structures. A Lewis structure also helps to make a prediction about the geometry of a molecule. The reason for learning to draw Lewis structures is to predict the number and type of bonds that may be formed around an atom. A Lewis structure is a graphic representation of the electron distribution around atoms.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
